Starting Date: 2022
Number of Partners: 5
Total Budget: 248K€
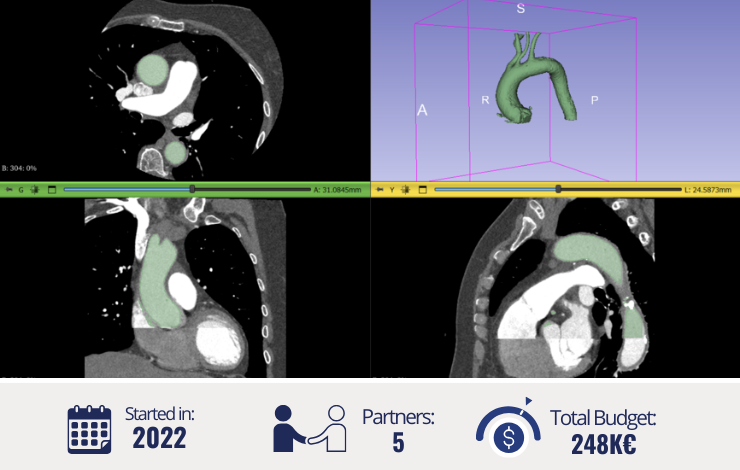
Thoracic aortic aneurysm (TAA) presents a complex clinical challenge due to its multifactorial aetiology and variable risk factors. Despite evolving surgical techniques and guidelines advocating for intervention in some instances, fundamental questions persist regarding disease prevention and diagnosis. Controversies surround the pathogenesis and risk of rupture, particularly as many patients experiencing aortic dissection have aortic diameters within the normal range. The project hypothesises that a personalised approach using computational modelling can benefit TAA treatment. Integrating patient-specific data from clinical exams, imaging, and ex-vivo measurements alongside advanced computational techniques like fluid-structure interaction (FSI) aims to understand better the relationship between TAA tissue structure and mechanical properties. This understanding will inform the development of mathematical models to assess hemodynamic factors, ultimately aiding clinical decision-making and risk stratification.
To address the deterministic limitations of current models, our project proposes introducing uncertainty quantification methods to account for biological variability. This innovative approach, analysing predictive responses from a statistical perspective and considering uncertainties in model inputs, aims to reduce the sources of uncertainty and improve the accuracy of predictions. Additionally, we aim to create a comprehensive metadata collection of clinical and imaging data to support the development of advanced computational tools. A key outcome is the development of a digital tool capable of modelling patient-specific TAA hemodynamics, providing evidence-based insights into disease progression and facilitating custom analyses for patients without immediate surgical indications. This novel approach seeks to optimise clinical decision-making by identifying patients who may benefit from early intervention and those who can safely continue outpatient follow-up, regardless of aortic diameter.
Visit the project page here.